Cellufine™ Sulfate
For concentration, purification and depyrogenation of virus, viral/microbial antigens and heparin-binding proteins
Advances in vaccines and clinical diagnostics have created an increasing demand for large volumes of highly purified and concentrated virus and viral or microbial antigens. Cellufine™ Sulfate affinity media is a simple, rapid and effective means for concentration, purification and depyrogenation of these important products.
Cellufine™ Sulfate eliminates cumbersome, time-consuming and potentially unsafe classical ultra-centrifugation and density gradient methods. It can also provide a significant improvement in concentration and purity. Cellufine™ Sulfate can reduce or eliminate the expense, ligand leakage and reproducibility problems associated with immobilized dextran sulfate, chondroitin sulfate or heparin.Elution of the bound product is affected through simple stepwise or gradient increases in ionic strength.
Features
- Affinity for a wide range of live, killed or disrupted viruses, viral or microbial antigens and heparin-binding proteins
- Closed column operation assures safety and product sterility
- Endotoxins do not bind, allowing a rapid and contaminant free depyrogenation
- Rigid, high-strength beads
- Autoclavable
Benefits
- More effective than ultracentrifugation at removing contaminants from culture media and host cells
- Avoids excessive product handling and safety concerns, particularly with viral preparations
- Simultaneous concentration and purification improve yield, reduce processing steps, time and costs
- Gentle binding and elution conditions provide high capacity and product yield
- Resists compression, providing rapid flow for high-speed processing, even in large columns, making it easily scalable
- Resistant to chemical depyrogenation with base and chemically sterilizable with formalin
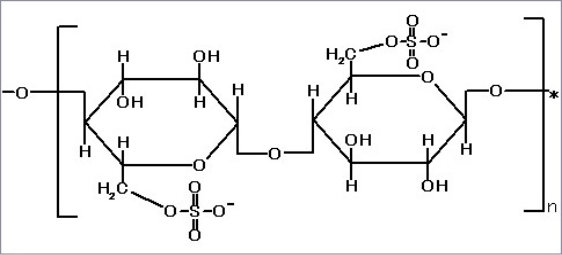
| Characteristics | |
|---|---|
| Support Matrix | Cellulose |
| Particle Size | ca. 40 – 130 µm |
| Particle Shape | Spherical |
| Gel Exclusion Limit | 3kD |
| Activated Group | Sulfate Ester |
| Total Sulfur | >700 µg/g dry |
| Protein Lysozyme : Hepatitis B Surface Antigen : |
>3 mg/ml 7 mg/ml |
| Environmental Resistance | Resistant to 0.1M NaOH,0.1 % of 37 % Formalin |
| Operating Pressure | <0.3 MPa |
| Autoclavable | In suspension at neutral pH; 30 min at 121 °C |
| Supplied | Suspension in 20 % Ethanol |
Flow Properties
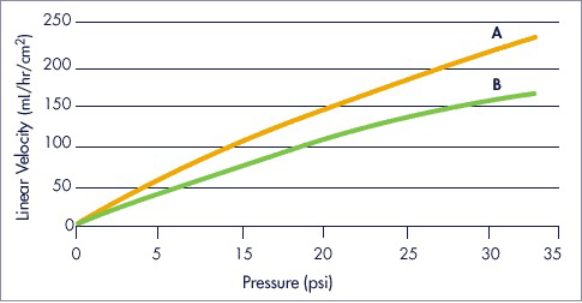
The nearly rigid properties of the spherical cellulose support matrix allow outstanding flow properties, particularly in large production columns.
- Column A
- 90 x 200 mm
- Column B
- 350 x 200 mm
Virus, Viral/Microbial Antigens
| Viruses | Viral/Microbial Agents |
|---|---|
|
|
| *These applications are covered by US and foreign process
patents. Please inquire regarding details and licensing arrangements. |
|
Table 1There are many applications of Cellufine™ Sulfate in the concentration or
purification of viral and microbial anifigensm proteins and viruses.
Example of purification of human coronavirus (OC43) with Cellufine™ Sulfate
Here is an example of purification of human coronavirus (OC43). Coronavirus has an envelope protein. So, Cellufine™ Sulfate is capable of adsorbing and purifying coronavirus.
Detailed technical data can be downloaded from the following.
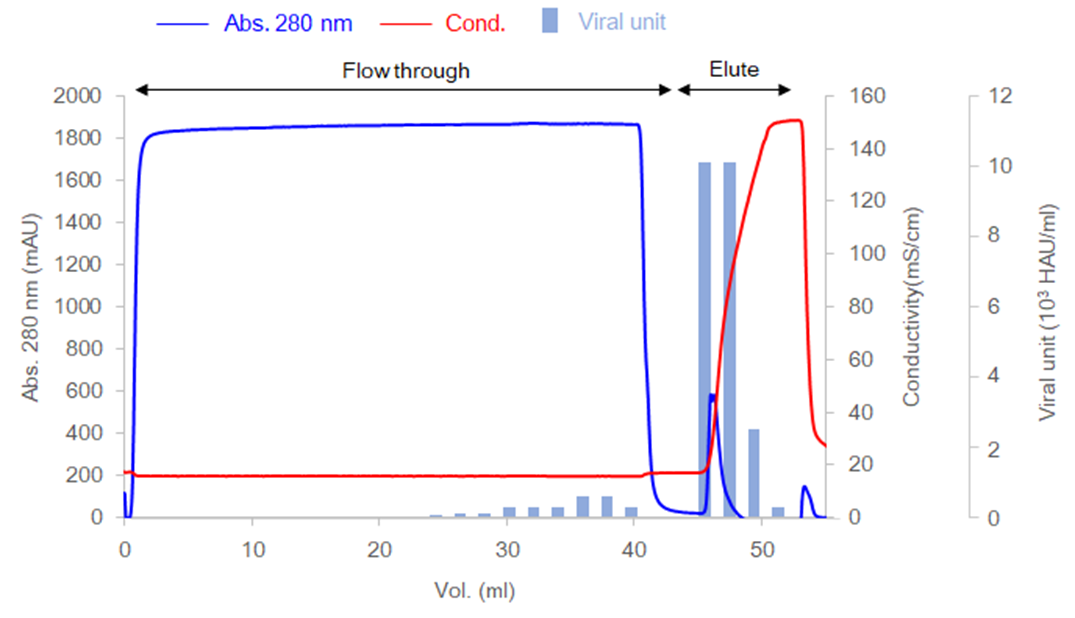
- Column
- I.D. 5 mm x height 15 mm (0.3 ml)
- Sample
- Vero cell lysate including BPL-inactivated virus
- Virus strain
- Human corona virus(HCoV) OC43
- Flow rate
- 0.3 ml/min (residence 1 min)
- Equilibration
- 10 mM sodium phosphate, 150 mM sodium chloride, pH 7.4
- Elution
- 10 mM sodium phosphate, 2 M sodium chloride, pH 7.4
Purification of Rabies Virus
The example in Figure 3 illustrates the high degree of concentration, purification and yields obtained with Cellufine™ Sulfate on typical viral preparations.
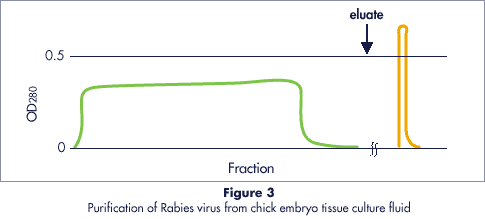
- Column
- 50 x 70 mm (140 ml)
- Buffer
- 0.01M Phosphate (pH 7.2)
- Eluant
- 1M NaCl/0.01M Phosphate (pH 7.2)
| Load | Eluate | |
|---|---|---|
| Volume (ml) | 4,200 | 50 |
| Virus titer | 32 | 4,096 |
| Protein (µg/ml) | 8.5 | 14 |
| Yield (%) | 100 | 152 |
| Purification factor | 79x | |
| Concentration factor | 126x |
Table 2Concetration and Purification of virus with Cellufine™ Sulfate
Purification of Influenza Virus
Hen’s egg allantoic fluid was loaded directly onto a 33.3 mL gel bed and 94.5%virus was recovered in the eluate fraction.
| Volume(ml) | Virus Titer | TCA-N µg/ml | Recovery(%) | Fold Purification | |
|---|---|---|---|---|---|
| Allantoic Fluid | 4200 | 77 | 337.1 | 100 | 1 |
| Wash | 6700 | 1 | 209.2 | 2.1 | - |
| Eluate | 170 | 1797 | 448.0 | 94.5 | 20.1 |
Table 3Purification of Influenza virus from hen's egg allantoic fluid
- Column
- 50 x 170 mm
- Buffer
- 0.01M Phosphate pH 7.4
- Wash
- 0.01M Phosphate pH 7.2 + 0.2M NaCl
- Elution
- 0.01M Phosphate pH 7.0 + 1.5M NaCl
Antigenic Protein Purification and Depyrogenation
Cellufine™ Sulfate is ideal for depyrogenating virus and other microbial extracts because it does not bind endotoxins. Figure 4 shows the purification of filamentous hemagglutinin (FHA) from the whooping cough bacterium Bordetella pertussis.
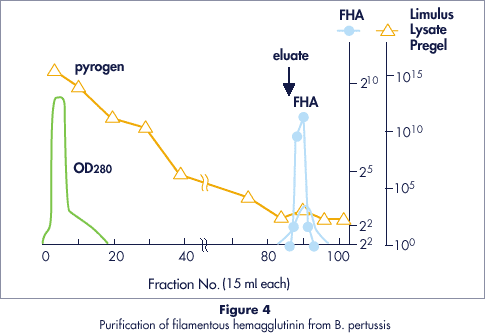
- Column
- 16 x 70 mm (20 ml)
- Sample
- 800 ml B. pertussis culture fluid
(endotoxin titer > 1015 by Limulus lysate test) - Buffer
- 0.01M Phosphate (pH 7.6)
- Eluant
- 1M NaCl/0.01M Phosphate (pH 7.6)
- FHA Yield
- 94%
- Purification Factor
- 20x
- Concentration Factor
- 28x (30 ml product)
- Endotoxin
- Below standard level by Limulus lysate, rabbit pyrogen and mouse toxicity tests
Protein Purification
Cellufine™ Sulfate mimics the affinity of heparin or dextran sulfate for many proteins. It can function as an affinity support for selected plasma proteins, cellular growth factors and lipases. Its capacity comparable to conventional heparin gels.
| Binding Proteins | Non-binding Proteins |
|---|---|
|
|
| *Binding and elution are extremely rapid and very fine separations can be generated in gradient mode. | |
Table 4
Purification of Partially Purified Casein Kinase II from Calf Thymus
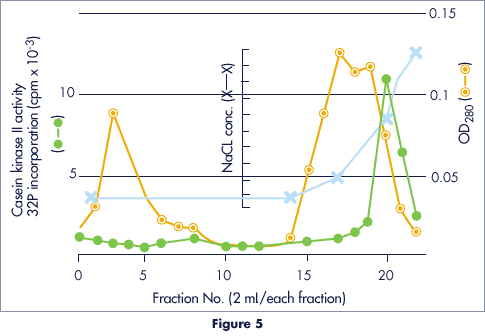
- Column
- 10 x 20 mm
- Sample
- 7 ml
- Buffer
- 50mM Tris-HCl (pH 7.9)
+ 50mM MgCl2
+ 0.1mM EDTA
+ 0.1mM PMS
+ 0.5mM DTT
+ 25 % glycerol - Eluant
- 0.05 – 1.0M NaCl in buffer
