Cellufine™ SPA-HCnew
Protein A media is selected as the first step of antibody purification in pharmaceutical company. In this step, it is important to purify high volume of culture supernatant in short time. So it is necessary to select high performance media described in below,
- 1. High dynamic binding capacity at high flow rate
- 2. Low back pressure at high flow rate
- 3. Reusability
Cellufine™ SPA-HC is the resin immobilized alkali resistance protein A ligand. The resin features low back pressure because of its crosslinking technology, and has high porosity in order to achieve high mass transfer of antibody.
The unique beads manufacturing technology obtained through experience of 40th years of
business
in chromatography makes the resin achieved high dynamic binding capacity at high flow
rate.
Cellufine™ SPA-HC was developed expecting used in manufacturing scale. So
current
equipment which you have can be availed achieving short of manufacturing time.
| Property | Characteristics |
|---|---|
| Ligand | Alkali-stable rProtein A affinity ligand produced in E.coli |
| Matrix | Highly cross-linked cellulose beads |
| Particle size | Average 70 µm diameter |
| Ligand coupling method | Coupling via formyl groups on resin |
| Flow velocity | ≧650 cm/h (0.3 MPa) with 10 cmID x 20 cmL Column in water at 24 ℃ |
| Dynamic binding capacity (DBC) | ≧ 70 mg /ml (DBC C10 with Polyclonal IgG) 1 |
| Recommend CIP solution | 0.1 M NaOH |
| Temperature stability | 4 - 50 ℃ No significant change in performance after 1-week storage. |
| Storage | 2 - 8 ℃ in 20 % (v/v) ethanol |
| Chemical stability | No significant change in performance after 1-week storage in 30 % (v/v) isopropanol, 20 – 70 % (v/v) ethanol, 8 M Urea, 6 M Guanidine-HCl or 0.1 M Acetic acid. |
| pH working range | 3-12 |
Features
- Coupled with an alkali resistance rProtein A ligand.
- Low back pressure due to its crosslinking technology achieving high porosity to facilitate high mass transfer of antibody with the immobilized affinity ligand.
- A unique smaller bead diameter manufacturing technology producing a resin with high dynamic binding capacity at high flow rates.
Cellufine™ SPA-HC has been developed for use with current equipment to achieve higher throughput and shorter manufacturing processes to accelerate production of antibody based biologics.
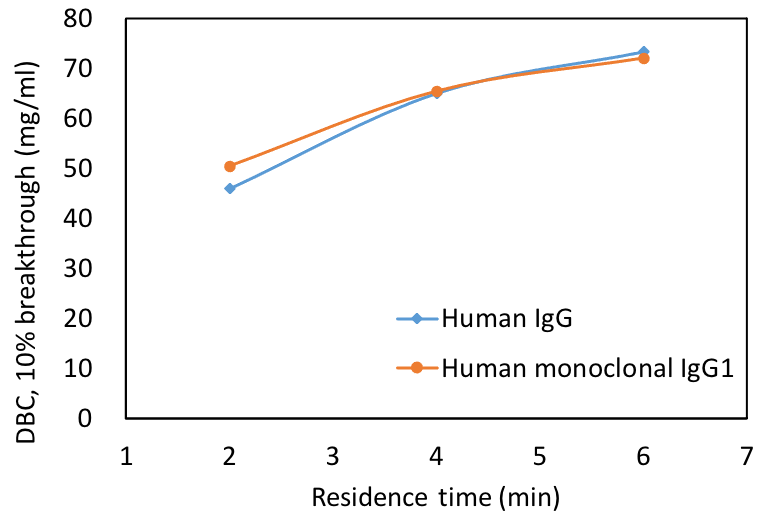
Excellent dynamic binding capacity
Cellufine™ SPA-HC shows high dynamic binding capacity with a human mAb sub-type 1 antibody (see Figure.1) indicating that this new resin can be used for efficient mAb purification workflows.
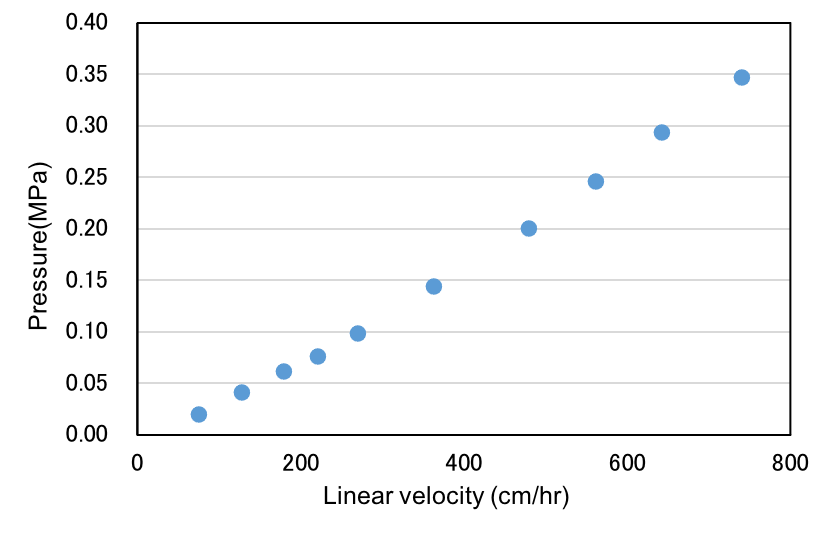
Low back pressure at high flow rate
As Cellufine™ SPA-HC is based on a highly cross-linked cellulose bead structure and is very adaptable for use in conventional manufacturing with large scale columns. Pressure-flow rate studies indicate that Cellufine™ SPA-HC developed back-pressures of <0.3MPa at 650 cm/h in a 10cmID x 20 cmL bed height (1.57 L) column.
The above data suggests that Cellufine™ SPA HC resin packed in a pilot process column can be operated at a residence time of 2.4min at < 0.3MPa with a flow rate of 650 cm/h and achieve a mAb capture loading of 50 mg/mL from a clarified CHO cell sample.
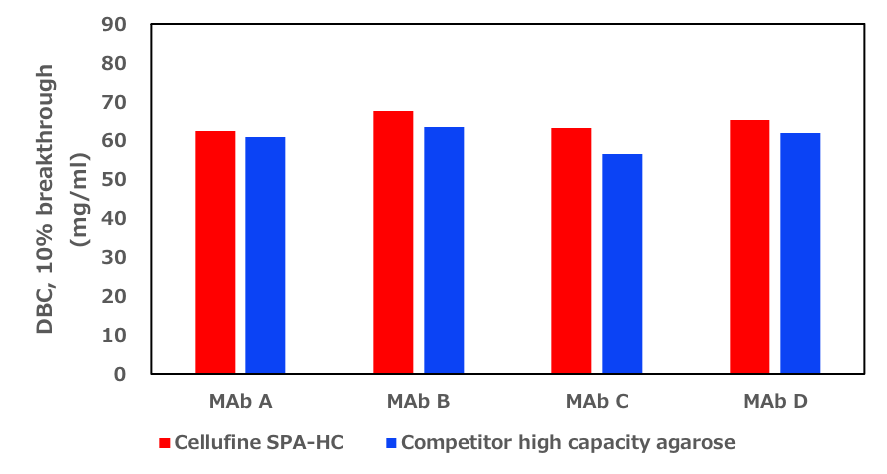
Robust performance not depend the sort of monoclonal antibody
Monoclonal antibodies with a range of isoelectric points (pI) and sub-types were used to investigate whether the molecular charge state had any impact on the dynamic binding capacity of the resin. The results indicate that Cellufine™ SPA-HC can adsorb antibodies with a wide range of pI’s and show higher DBC capacity compared to an 85 µM high capacity agarose competitive resin. A summary is shown in Figure 3, below.
Excellent reusability
Cleaning in place (CIP) with base is routinely applied during cycles of re-use of
rProtein
affinity resins. Cellufine™ SPA-HC has been evaluated for 0.1 M NaOH base CIP for up to
150
cycles of re-use. Results are summarized in Figure 4 below.
The data shows high
retention
(>95%) of DBC after 150 cycle 0.1M NaOH CIP. The capacity at cycle zero was 75 mg/mL and
changed
to 72 mg/mL at the end of the study.
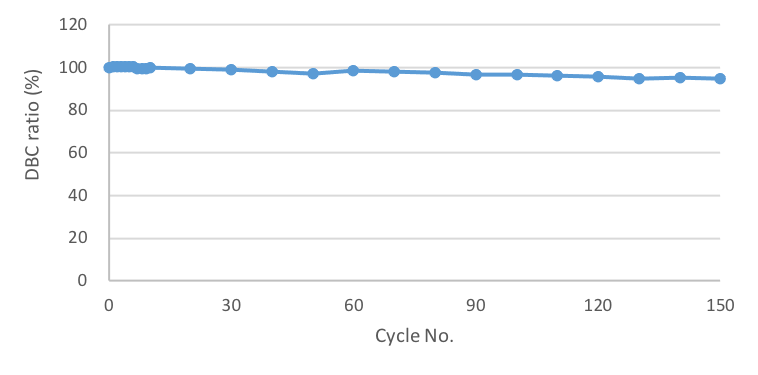
This figure shows remaining dynamic binding capacity as a ratio. 10 % breakthrough DBC was 76 mg/mL at the first time. After 150 cycle alkaline attachment, DBC changed 72 mg/mL adsorption. It means that 95% activity was remaining after this study finished.
- CellufineTMSPA-HC
