Cellufine™ MAX S, Q, CM, DEAE, GS
High Flow Rate, High Binding Capacity
Cellufine™ MAX is the new, high-flow, Cellufine™ media. JNC’s advanced cross-linking technologies have created more robust base beads operable at high flow and pressure. Further, Cellufine™ MAX ion exchange (IEX) media are made using surface modification techniques that dramatically increase ligand availability, which translates to higher dynamic binding capacities. Cellufine™ MAX IEX media are offered in six products, including both anion and cation chemistries.
Cellufine™ MAX Base Resin
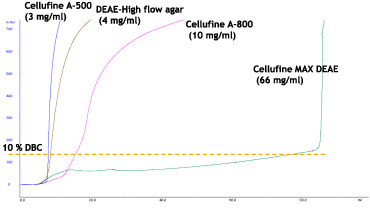
Cellulose, natural polysaccharide, possesses unique crystalline molecular structure differing from non-crystalline polysaccharides such as agarose. Thus Cellufine™ has unique pore structure as shown in the pictograph (Fig. 1). The new Cellufine™ MAX series offers the largest pore size of all Cellufine™ chromatography media. The benefit of such pore size in Cellufine™ MAX IEX media provides superior strength and excellent mass transfer. This is seen in the break-through curves for thyroglobulin, a very large protein (Fig. 2).
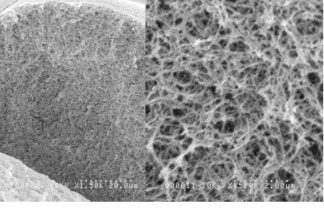
Partial Structure of Cellufine™ MAX IEX Media
Ligand structure for Cellufine™ MAX IEX media are described in Fig. 3. S, Q, CM and DEAE are correspondingly strong cation, strong anion, weak cation and weak anion exchangers. Two sub-types, h and r, are available for Cellufine™ MAX S and Q.
The differences between X-h and X-r type Cellufine™ MAX strong ion exchange media (X) are due to the design of the media. The X-h type is designed for higher binding capacity than the X-r type by optimizing the ligand content and dextran scaffold.
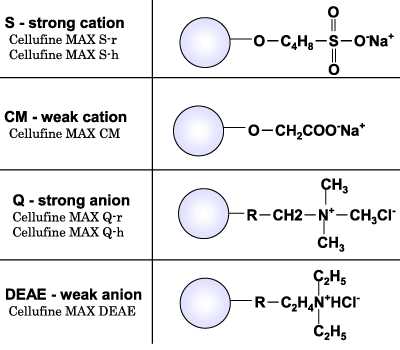
Characteristics of Cellufine™ MAX IEX Media
The basic characteristics of Cellufine™ MAX IEX media are shown in Table 1. All Cellufine™ MAX IEX media are based on 90 μm (average) highly cross-linked cellulose beads, which are surface-modified with dextran. Cellufine™ MAX IEX media are designed for use in bio-pharmaceuticals purification processes.
| Type | MAX CM | MAX S-r | MAX S-h | MAX DEAE | MAX Q-r | MAX Q-h | |
|---|---|---|---|---|---|---|---|
| Matrix | Cross-linked cellulose with dextran scaffold | ||||||
| Particle size (μm) | 40 - 130 | ||||||
| Ligand | CM | S | S | DEAE | Q | Q | |
| Ion exchange capacity (meq / ml-gel) | 0.09 - 0.22 | 0.09 - 0.21 | 0.10 - 0.22 | 0.12 - 0.22 | 0.10 - 0.20 | 0.13 - 0.22 | |
| 10% DBC(mg/ml) | Lysozyme | 220 | 144 | 191 | |||
| BSA | 197 | 141 | 225 | ||||
| human-γ-globulin | 104 | 131 | 216 | 108 | 74 | 135 | |
| pH stability | 2 -13 | 2 -13 | 3 -14 | 2 -12 | 2 -12 | 2 -12 | |
| Storage | 20% Ethanol | ||||||
Pressure-flow Properties of Cellufine™ MAX IEX Media
Cellufine™ MAX IEX media enable high-flow operation, which is essential to efficient purification of bio-pharmaceuticals.
The figures below show pressure-flow velocity curves of Cellufine™ MAX IEX media in a 30 cm column with a 20 cm bed height (Fig. 4). All Cellufine™ MAX IEX media are operable at practical flow velocities (500 cm/h) and pressures.
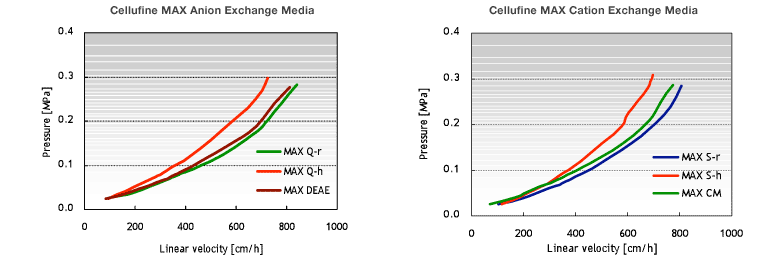
- Column
- 30 cm I.D. x 20 cm L
- Mobile phase
- Pure Water at 24 ºC
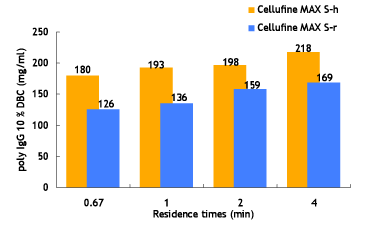
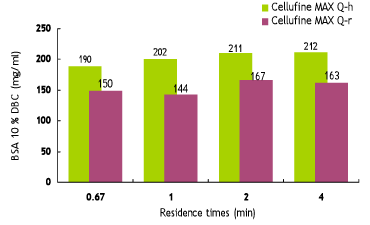
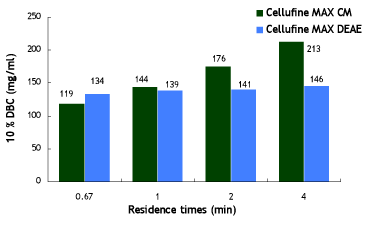
Dynamic Binding Capacities of Cellufine™ MAX IEX Media
Efficient mass-transfer characteristics of Cellufine™ MAX IEX media translate to superior dynamic binding capacities (DBC). Figure 5 to 7 show DBC of model proteins at different residence times for Cellufine™ MAX IEX media. All Cellufine™ MAX IEX media are stable over a range of residence times.
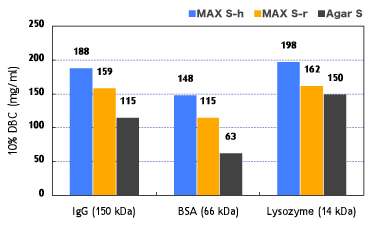
Fig. 8 shows that Cellufine™ MAX S exhibits superior dynamic binding performance across a range of protein characteristics to competitive media.
These unique characteristics of Cellufine™ MAX IEX media make it suitable for use in up-stream as well as to down-stream steps in bio-pharmaceuticals purification.
Cellufine™ MAX Cation Exchange Media
- Column
- 5 mm ID×50 mm L
- Sample
- human polyclonal IgG (1 mg/ml)
- Buffer
- 10 mM Acetate-50 mM NaCl (pH 4.3)
Cellufine™ MAX Anion Exchange Media
- Column
- 5 mm I.D. x 100 mm L
- Sample
- BSA (1 mg/ml)
- Buffer
- 50 mM Tris-HCl (pH 8.5)
Cellufine™ MAX Weak ion Exchange Media
- Column
- 5 mm ID x 50 mm L
- Sample
- human polyclonal IgG (1 mg/ml)
BSA (1 mg/ml) - Buffer
- 10 mM Acetate (pH 5.6) for IgG
Tris-HCl (pH 8.5) for BSA
Cellufine™ MAX Cation Exchange Media
- Polyclonal IgG
- 10 mM Acetate (pH 4.3) - 50 mM NaCl
- BSA
- 10 mM Acetate (pH 4.3) - 50 mM NaCl
- Lysozyme
- Tris-HCl (pH 9.5)
Model Proteins Separation Performance for Cellufine™ MAX IEX Media
Cellufine™ MAX IEX media are optimized for high adsorption and high resolution. Model protein separation with MAX S-h and MAX CM (Strong Cation vs. Weak Cation) is demonstrated in Fig. 9 and 10.
Cellufine™ MAX Cation Exchange Media
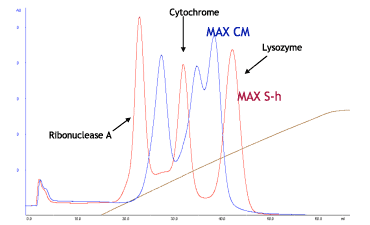
- Column
- 6.6 mm ID×50 mm L
- Buffer A
- 10 mM phosphate buffer (pH 7)
- Buffer B
- 10 mM phosphate (pH 7) + 1 M NaCl
(0→50 % linear gradient) - Flow rate
- 0.86 ml/min (residence time: 2min)
- Proteins
- Ribonuclease A (5 mg/ml),
Cytochrome C (2.5 mg/ml),
Lysozyme (1.5 mg/ml) - Injection volume
- 1.5ml
Cellufine™ MAX Anion Exchange Media
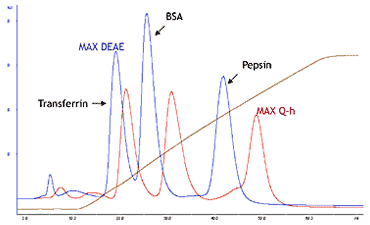
- Column
- 6.6 mm ID×50 mm L
- Buffer A
- 50 mM Tris-HCl (pH 8.5)
- Buffer B
- 50 mM Tris-HCl (pH 8.5) - 1 M NaCl
(0→75 % linear gradient) - Flow rate
- 0.86 ml/min (residence times 2 min)
- Proteins
- Transferrin (5 mg/ml),
BSA (10 mg/ml),
Pepsin (5 mg/ml) - Injection volume
- 1.5 ml
Chemical Stability and Cleaning-In-Place
Cellulose is well-known as a natural product having chemical and physical stability. Thus, since Cellufine™ is derived from cellulose, it also is stable to chemicals, caustic and acidic solutions. CIP of all Cellufine™ MAX IEX media can be carried out with 0.5 M NaOH solution. Used media should be stored in 20 % ethanol at 2 - 25 ºC after cleaning.
Cellufine™ MAX GS has been developed as a new strong cation chromatography media, with optimized ligand density. Cellufine™ MAX GS shows a superior performance for aggregate removal from therapeutic Mabs.
Characteristics of Cellufine™ MAX GS Media
The basic characteristics of Cellufine™ MAX GS are shown in Table 1. The base matrix for Cellufine™ MAX GS is 90 μm (average) highly cross-linked cellulose beads, the same as other Cellufine™ MAX IEX media.
| Matrix | Highly Cross-linked Cellulose |
|---|---|
| Particle size | 40~130 μm |
| Ligand type | -R-SO3-Na+ |
| Ion Exchange Capacity (m mol / ml) | 0.09〜0.15 |
| Lysozyme adsorption capacity (mg / ml) | ≧ 100 |
| Polyclonal IgG 10% DBC (mg / ml) | ≧ 70 |
| Operating pressure | < 0.3 MPa |
| pH stability | pH 2 ~ 13 |
Pressure-Flow Properties of Cellufine™ MAX GS
Figure 1 shows pressure-flow velocity curves of Cellufine™ MAX GS in 30 cm I.D. column with 20 cm bed height. Cellufine™ MAX GS has excellent flow properties and is applicable for process production.
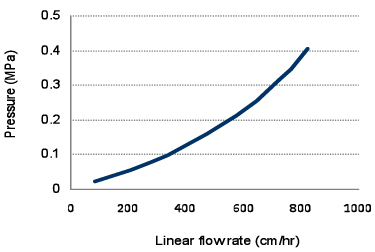
- Column
- 30 cm I.D. x 20 cm L
- Mobile Phase
- Pure water (24℃)
Model Protein Separation Performance for Cellufine™ MAX GS
Cellufine™ MAX GS is highly effective aggregates from IgG monomer by NaCl or pH gradient. Figure 2 shows the comparison of Cellufine™ MAX GS with S agarose media for monomer /aggregate separation of polyclonal IgG (Figure 2a) and monoclonal antibody (Figure 2b) by NaCl gradient elution. Cellufine™ MAX GS has been shown to be applicable an effective media for aggregate removal from Mabs.
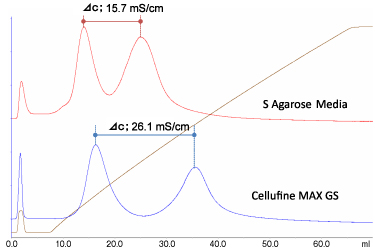
- Poly IgG
- Thermal and acid stressed Poly IgG
- Buffer
- Acetate (pH5.0), 50mM →1 M NaCl Poly IgG
- injection
- 1 ml
- Poly IgG conc
- 2 mg/ml
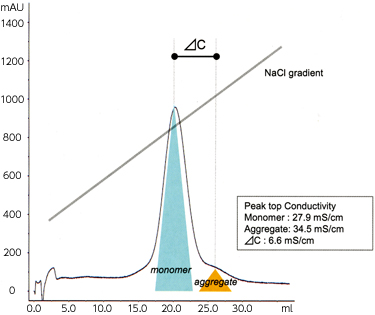
- Column
- 5 mm ID × 50 mm L
- Buffer
- Citrate Buffer (pH 5.0)
- NaCl Gradient
- 0.2→0.5 M
- Mab Injection
- 1 ml
- Flow rate
- 0.66 ml/min
Dynamic Binding Capacities of Cellufine™ MAX GS
Efficient mass-transfer characteristics of Cellufine™ MAX GS translate to superior dynamic binding capacities (DBC). Figure 3 shows DBC of Poly IgG at various residence times. Cellufine™ MAX GS is suitable for use in down-stream steps in antibody purification.
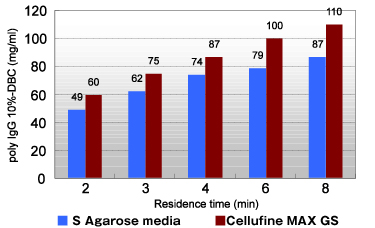
- Conditions
- Column
- φ5 mm×5 cm L
- Poly_IgG Concentration
- 1 mg/ml
- Adsorption Buffer
- 10 mM Acetate (pH 5.0) + 50 mM NaCl
- Cellufine™ MAX S-r , S-h
- Cellufine™ MAX GS
- Cellufine™ MAX Q-r , Q-h
- Cellufine™ MAX CM
- Cellufine™ MAX DEAE
